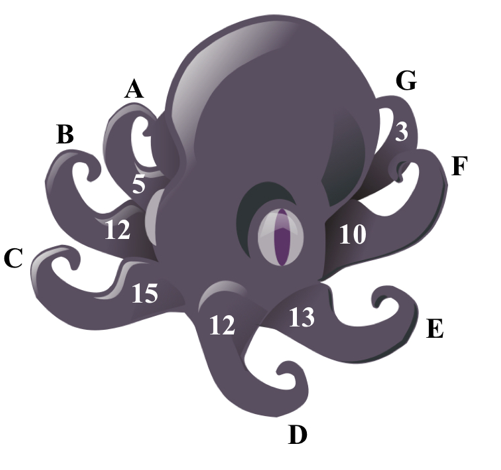

Consider an octoopus with seven limbs (labeled A-G), separated brom each other by a selectively permeable membrane that only allows water to flow between each arm. At steady state equuilibrium, each arm has a beginning osmolarity of 300 mOsm/L, and volumes as indicated in the figure.
If 2200 ml of a 1.5M solution of CaCl2 (i = 2.1) is injected into limb "E", what would be the new steady state volumes for all the limbs following equilibrium after osmosis occurs?